Results for Back Titration
Results for Synthesis of MCP
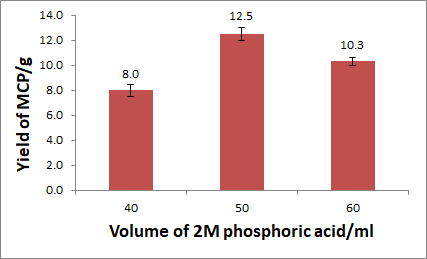
Figure 1: Percentage yield of MCP synthesized from different volumes from 2M phosphoric acid
Yield of MCP/g
The figure above shows that 50 ml of 2M phosphoric acid is the ideal volume of phosphoric acid required to obtain the greatest yield of MCP. Any volume lower or higher than this significantly reduces the amount of yield obtained. Since the mass of blood cockle shells was kept constant throughout all the experiments, it can be deduced that the optimum ratio for the synthesis of MCP is 5g of cockel shells to 50ml of 2M phosphoric acid.
Results for Effect of Heating on Yield of MCP
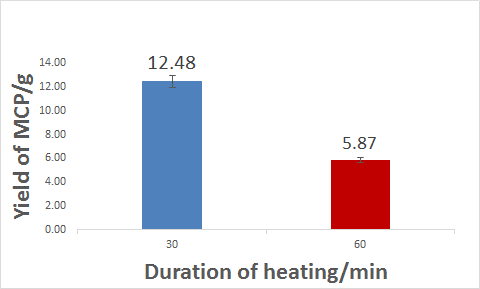
Figure 2.1: Effect of duration of heating on yield of MCP
As duration of heating increases from 30 min to 60 min, the yield of MCP decreases significantly. This could be attributed to side reactions occurring when heating is prolonged, leading to formation of insoluble phosphates such di-calcium phosphate and tri-calcium phosphate
Figure 2.2: Residue obtained after 60 min (left) and 30 min (right) of heating
This is evident from the amount of residue recovered after the filtration (figure 2.2). There was hardly any residue (0.34g) obtained from 30 min of heating. In contrast, as much as 3.08 g of residue was obtained from 60 min of heating.
Results for Analysis of MCP
|
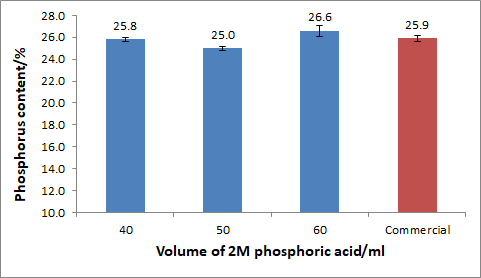
Figure 3.1: Percentage Phosphorus content in MCP synthesized from different volumes from 2M phosphoric acid
% of Calcium ContentThe figure above which shows the analysis of phosphorus in the MCP synthesised from various volumes of phosphoric acid shows that the percentage of phosphorus content ranges from 25-27%. This is comparable to the amount of phosphorus content in commercial MCP which ranges from 24-25%. Therefore, it can be concluded that the phosphorus content in the MCP synthesised is slightly higher than that of commercial MCP.
|
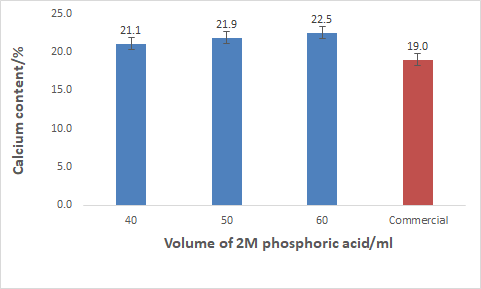
Figure 3.2: Percentage Calcium content in MCP synthesized from different volumes from 2M phosphoric acid
% of Phosphorus ContentThe figure above which shows the analysis of calcium in the MCP synthesised shows that the percentage of calcium ranges from 21-23%. The reported percentage of calcium from commercial MCP ranges from 18-20%. Thus, the MCP synthesised from blood cockle shells has a slightly higher calcium content than that of commercial MCP.
|
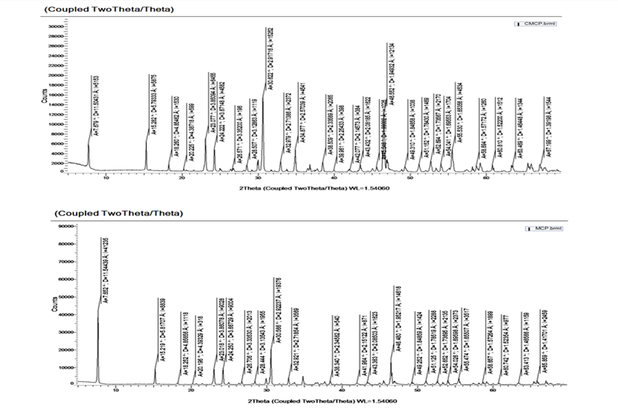
Figure 3.1: XRD(X-ray diffraction) results of Commercial MCP and MCP synthesised
Commercial MCP
MCP synthesised from blood cockle shells
MCP synthesised from blood cockle shells
Figure 3.2 SEM(Scanning electron microscope) which show a triclinic lattice structure
|
Commercial MCP
|
MCP from blood cockle shells
|
From the 4 figures above, it can be seen that the MCP synthesised has a similar shape which corresponds with the triclinic lattice structure of the commercial MCP. Hence, it can be concluded that the crystals synthesised is indeed MCP. Furthermore, from X-ray diffraction, it can be seen that the peaks and interdistance layer for the synthesised MCP and commercial MCP are very similar. As the position of the diffraction peaks are determined by the distance between parallel planes of atoms, it can also be concluded that the MCP synthesised is exceedingly similar to that of commercial MCP in terms of structure and shape.
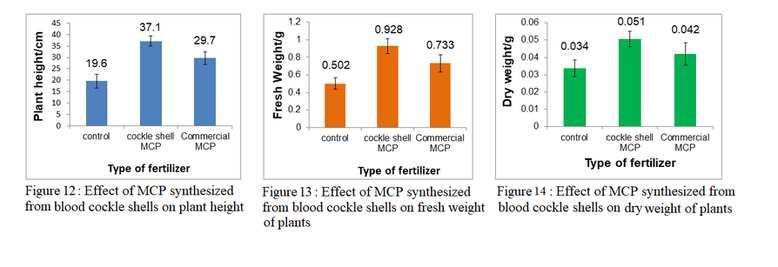
Results for Evaluating MCP as a potential Plant Fertilizer
As can be seen from figure 12-14, MCP synthesized from blood cockle shells enhances plant growth, resulting in an increase of 89.3% in plant height, 84.9% in fresh weight and 50.0% in dry weight as compared to control. This is an indication that the MCP synthesized from blood cockle shells is able to provide nutrients, namely calcium and phosphorus to the plants.